Chemistry: What is a sigma bond (σ bond)? What is a pi bond (π bond)?
The tutor briefly describes σ bond and π bond.
Sigma bonds and pi bonds are types of chemical bonds that hold molecules together.
In a sigma bond, the pair of electrons is found between the two atoms involved. The atoms, with their positively-charged nuclei, are attracted to the electrons. Therefore, while the electrons remain between them, in the sigma bond, the two atoms will stay together. A single bond is typically a sigma bond.
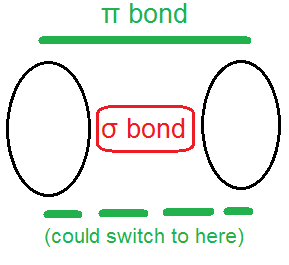
With a pi bond, the pair of electrons resides in a cloud parallel to the sigma bond, but offset from it. Rotation is not possible around a pi bond without breaking it. A pi bond can switch between “poles”: each pi bond has two possible locations where its electrons might be found.
Typically, a double bond indicates a sigma bond together with a pi bond.
The drawing below depicts both a sigma bond (σ bond) and a pi bond (π bond). Note it shows only one π bond. The other one, if present, would be parallel, but in front of (and behind) the diagram.
HTH:)
Source:
Mortimer, Charles E. Chemistry, 6th ed. Belmont: Wadsworth, Inc., 1986.
Jack of Oracle Tutoring by Jack and Diane, Campbell River, BC.
Leave a Reply
You must be logged in to post a comment.